

IBT specializes in delivering top-notch reagents tailored for research and development purposes.

Development by providing comprehensive, innovative and tailored solutions.

In biomedical innovation, helping our clients navigate the complex preclinical landscape with confidence.

By promoting transparency, collaboration, data integrity, and scientific excellence.

To make impactful breakthroughs that transform lives.

To our clients in their journey of biomedical research and innovation.

To address the unique challenges of each project.
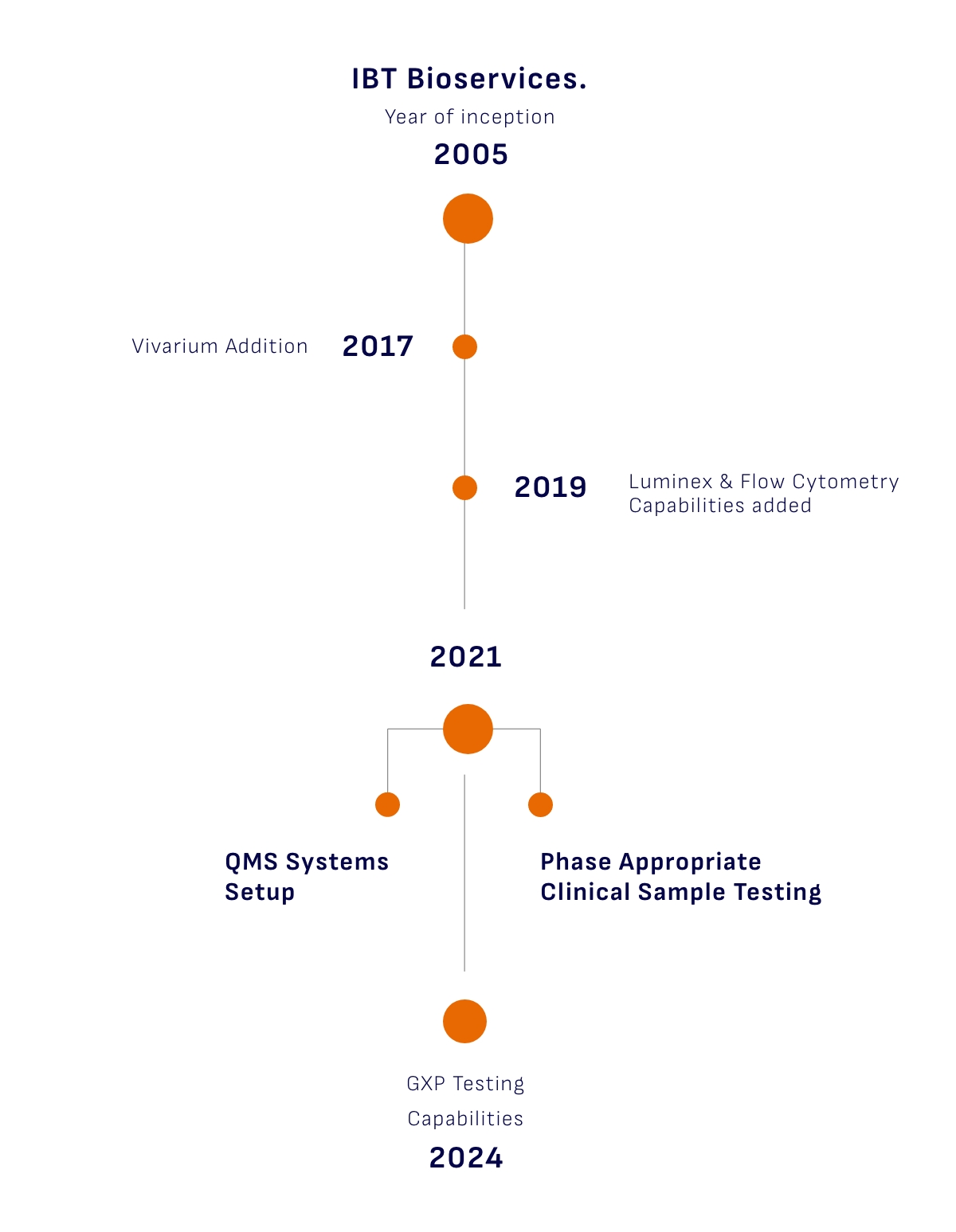
Our purpose is to serve as a trusted partner to our client in their journey of biomedical research and innovation. We are committed to enabling the successful translation of novel therapies from the research stage to clinical application. Our goal is to help our client bring groundbreaking treatments to patients, marking significant contributions to the advancement of healthcare.





For concerns regarding quality issues unrelated to temperature, customers may request a return or exchange within 7 days of receipt. Our quality control team will assess the returned items, and upon confirmation of the issue, a decision will be made to replace the product or issue a refund.
If customers mistakenly order the wrong reagent, we may consider exchanges under certain conditions. Items that have not yet been shipped or unopened packages can be exchanged. The customer will be responsible for shipping costs.
To initiate a return or exchange, customers must contact our Product Representatives team at services@IBTBioservices.com.
Note: All returns/exchanges must be shipped back within 7 days of notifying our product representatives about the issue. We reserve the right to refuse returns that do not meet the specified conditions outlined in this policy.
IBT’s capabilities include BSL-2 testing. We offer BSL-3 and –4 testing capabilities through our partners.
To explore potential career opportunities with us please navigate to the Careers section of our webpage where you will find a list of current job openings, each accompanied by detailed job descriptions and application instructions. IBT is an equal opportunity employer.